Oocyte maturation and fertilization of marine invertebrates (Fertilization process of A. aranciacus, courtesy of L. Santella, Stazione Zoologica Anton Dohrn).
Background of the study
Reproduction is one of the fundamental subjects in the field of developmental
biology. In the case of most animals, fertilization process is involved
in the reproduction. In our laboratory, we study the mechanism of the fertilization
especially how the oocytes obtain the ability for re-initiation of meiosis
and further development after fertilization, how the fertilizing sperm
enter the oocyte, and how the activation of the oocytes takes place by
fertilizing sperm. Marine invertebrates, such as echinoderms, mollusks,
ascidians, and hydrozoas are our favorite materials. Around our Marine
Biological Station, it is easy to obtain a plenty of these fresh animals.
Generally, the oocytes dissected from these gametes are easy to fertilize
and this process is easy to observe under a microscope.
Some of our experimental materials


Ciona savignyi (left panel) and several species of mollusks (rignt panel)
Recent studies
Our main interest now is calcium signaling during oocyte maturation and fertilization. We measure the calcium changes using fluorescent calcium indicators with confocal microscopy, high sensitive CCD camera or photomultipilers. Our research subjects in progress are 1. The role of calcium ions and calcium dynamics during oocyte maturation in starfish oocytes, and 2. The purification and characterization of egg activating substance (sperm factor) from ascidian sperm.
Change of calcium ions in ascidian (Ciona savignyi) oocytes after fertilization
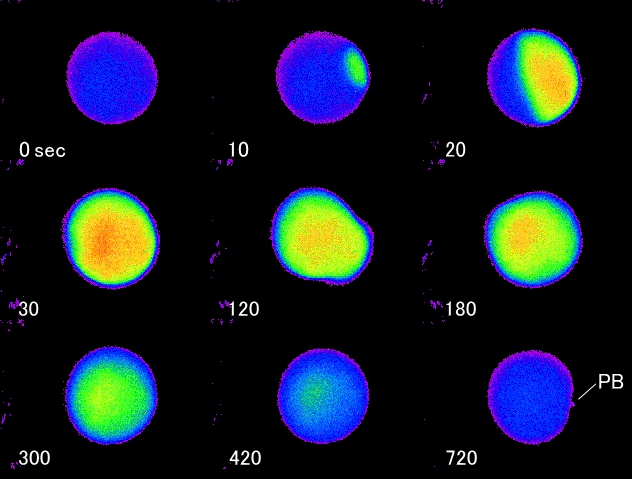
Fertilization of Ciona oocyte takes place at metaphase I of meiotic divisions
(0 sec). Calcium wave started at the point where the fertilizing sperm
entered and it propagated across the oocyte to the opposite side of the
sperm entry point (10, 20, 30 sec). Two minutes after fertilization, egg
contraction that courses the cytoplasmic segregation was observed (120
sec). Several calcium oscillations followed after the first calcium wave
(180, 300, 400 sec) and the first polar body protruded at 12 min after
fertilization (720 sec, PB). Free calcium level was low in the oocyte for
5 min after protrusion of the first polar body. Then second set of calcium
oscillations started and it stopped when second polar body protruded at
30 min after fertilization.
Recent Publications
Doticon_grn_NEW.pngSoluble sperm extract specifically recapitulates the
initial phase of the Ca2+ response in the fertilized oocyte of P. occelata
following a G-protein/ PLCβ signaling pathway.
Nakano T, Kyozuka K.
Zygote. 2014 Oct 16:1-15.
Doticon_grn_NEW.pngIntracellular calcium signaling in the fertilized eggs
of Annelida.
Nakano T, Deguchi R, Kyozuka K.
Biochem Biophys Res Commun. 2014 Aug 1;450(3):1188-94. Review.
Guanine nucleotides in the meiotic maturation of starfish oocytes: regulation
of the actin cytoskeleton and of Ca(2+) signaling.
Kyozuka K, Chun JT, Puppo A, Gragnaniello G, Garante E, Santella L.
PLoS One. 2009 Jul 20;4(7):e6296. PMID:19617909
Nitric oxide (NO) increase at fertilization in sea urchin eggs upregulates
fertilization envelope hardening.
Mohri T, Sokabe M, Kyozuka K.
Dev Biol. 2008 Oct 15;322(2):251-62. Epub 2008 Jul 29. PMID:18694744
Actin cytoskeleton modulates calcium signaling during maturation of starfish
oocytes.
Kyozuka K, Chun JT, Puppo A, Gragnaniello G, Garante E, Santella L.
Dev Biol. 2008 Aug 15;320(2):426-35. Epub 2008 Jun 6. PMID:18599031
Novel two-step Ca2+ increase and its mechanisms and functions at fertilization
in oocytes of the annelidan worm Pseudopotamilla occelata.
Nakano T, Kyozuka K, Deguchi R.
Dev Growth Differ. 2008 Jun;50(5):365-79. Epub 2008 Apr 28. PMID:18445067
Two phases of calcium requirement during starfish meiotic maturation.
Tosuji H, Seki Y, Kyozuka K.
Comp Biochem Physiol A Mol Integr Physiol. 2007 Jun;147(2):432-7. Epub
2007 Jan 30. PMID:17317251
Serotonin stimulates [Ca2+]i elevation in ciliary ectodermal cells of echinoplutei
through a serotonin receptor cell network in the blastocoel.
Katow H, Yaguchi S, Kyozuka K.
J Exp Biol. 2007 Feb;210(Pt 3):403-12. PMID:17234609
Increase in intracellular cAMP is a prerequisite signal for initiation
of physiological oocyte meiotic maturation in the hydrozoan Cytaeis uchidae.
Takeda N, Kyozuka K, Deguchi R.
Dev Biol. 2006 Oct 1;298(1):248-58. Epub 2006 Jun 27. PMID:16884710
Modulation of calcium signalling by the actin-binding protein cofilin.
Nusco GA, Chun JT, Ercolano E, Lim D, Gragnaniello G, Kyozuka K, Santella
L.
Biochem Biophys Res Commun. 2006 Sep 15;348(1):109-14. Epub 2006 Jul 17.
PMID:16875665
NAADP and InsP3 play distinct roles at fertilization in starfish oocytes.
Moccia F, Nusco GA, Lim D, Kyozuka K, Santella L.
Dev Biol. 2006 Jun 1;294(1):24-38. Epub 2006 Mar 20. PMID:16545362
NAADP triggers the fertilization potential in starfish oocytes.
Moccia F, Lim D, Kyozuka K, Santella L.
Cell Calcium. 2004 Dec;36(6):515-24. PMID:15488601
The M-phase-promoting factor modulates the sensitivity of the Ca2+ stores
to inositol 1,4,5-trisphosphate via the actin cytoskeleton.
Lim D, Ercolano E, Kyozuka K, Nusco GA, Moccia F, Lange K, Santella L.
J Biol Chem. 2003 Oct 24;278(43):42505-14. Epub 2003 Jul 16. PMID:12867432
NAADP+ initiates the Ca2+ response during fertilization of starfish oocytes.
Lim D, Kyozuka K, Gragnaniello G, Carafoli E, Santella L.
FASEB J. 2001 Oct;15(12):2257-67.PMID:11641253
Breakdown of cytoskeletal proteins during meiosis of starfish oocytes and
proteolysis induced by calpain.
Santella L, Kyozuka K, Hoving S, Munchbach M, Quadroni M, Dainese P, Zamparelli C, James P,
Carafoli E.
Exp Cell Res. 2000 Aug 25;259(1):117-26. PMID:10942584
Nicotinic acid adenine dinucleotide phosphate-induced Ca(2+) release. Interactions
among distinct Ca(2+) mobilizing mechanisms in starfish oocytes.
Santella L, Kyozuka K, Genazzani AA, De Riso L, Carafoli E.
J Biol Chem. 2000 Mar 24;275(12):8301-6. PMID:10722659
Cortical granule translocation during maturation of starfish oocytes requires
cytoskeletal rearrangement triggered by InsP3-mediated Ca2+ release.
Santella L, De Riso L, Gragnaniello G, Kyozuka K.
Exp Cell Res. 1999 May 1;248(2):567-74. PMID:10222148
Injection of sperm extract mimics spatiotemporal dynamics of Ca2+ responses
and progression of meiosis at fertilization of ascidian oocytes.
Kyozuka K, Deguchi R, Mohri T, Miyazaki S.
Development. 1998 Oct;125(20):4099-105. PMID:9735370
